
Does the LoD cutoff + the TIMI score = a NICE approach to rule-out a major adverse cardiac event in the emergency department?
The literature is abundant with accelerated diagnostic pathways (ADPs) for early decision making in emergency department (ED) patients presenting with chest pain (1-10). By in large, this growth in the various ADPs has been possible due to the introduction of high-sensitivity cardiac troponin (hs-cTn) testing. Published data, nearly a decade ago, demonstrated the utility of how the more analytically sensitive and precise hs-cTn assays can lead to an earlier diagnosis and provide important prognostic information for patients presenting with chest pain to the ED (11-13). Yet despite all the data, no ADP has proven to provide better patient outcomes than the current standard of care. This is because these ADPs have not necessarily improved diagnostic performance or capture all the outcomes of interest for the physicians practicing in the ED (7-10). In fact, one could go further, and simply state that despite all the improvements made to hs-cTn, it is still just a diagnostic “test” and like any test there are errors, interferences and other variables that may affect its analytical performance and thereby affect its clinical performance (14). This point cannot be under-emphasized considering the bench mark most ED physicians consider safe for ruling-out major adverse cardiovascular events (MACE) is a sensitivity >99% (15).
The improvements to the hs-cTn tests now allow these assays to measure cTn concentrations in the majority of healthy individuals. However, the cutoff chosen for the majority of ADPs for ruling-out an event is the limit of detection (LoD), essentially reducing this test to a binary detected/not detected response. This approach is problematic on several fronts (16-20):
- First, there is no requirement that laboratories use the same lower reportable limit for the hs-cTn assays (thereby a “undetectable” result may be different between laboratories and studies);
- Second, there are no quality procedures in place to monitor test performance at the lower limit of reporting (thus the quality of measurements are unknown);
- Third, there are no accepted standard operating procedures to prevent drift of the assay at the lower reportable limit when changing lots of reagents (thus no guidance for unacceptable lots);
- Fourth, the repeatability and acceptable error estimates at the lower reportable limit can readily re-classify patients (thus a repeat measurement could change the decision to rule-out).
Reliance on more than just an undetectable hs-cTn result is, therefore, paramount and thus the coupling of this test with other validated tools. One such tool that has been used in ADPs is the Thrombolysis In Myocardial Infarction (TIMI) score (21). The latest 2016 National Institute for Health and Care Excellence (NICE) high-sensitivity troponin rule-out strategy suggests that physicians “consider performing a hs-cTn test only at presentation to rule-out non-ST elevation myocardial infarction (NSTEMI) if the test is below the limit of detect (LoD)’ and “the patient is low-risk as indicated by a validated tool” (22). Carlton and colleagues study (publication in the journal Heart) evaluated such an approach in >3,000 patients with hs-cTnT (Roche Diagnostics), hs-cTnI (Abbott Diagnostics), and TIMI scores (22). Their findings that a TIMI score of 0 coupled with the LoD cutoff, yielded a sensitivity of 99.5% (hs-cTnT) and 98.9% (hs-cTnI); supportive of the recommendation by NICE (22). This approach would limit this ADP to only patients <65 years of age (as age ≥65 years yields a TIMI score of 1). If, however, one accepts a lower sensitivity (range, 98.4–98.9%) by using a TIMI score ≤1, then this approach might be useful for those 65 years and older.
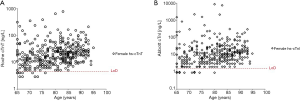
Intriguingly, neither the NICE guideline, nor other proposed rule-out pathways have incorporated sex as a variable (1,3-6,21,22); though there is data (for and against) that different cutoffs should be used for the sexes to rule-in MI (23,24). We have recently assessed the presentation hs-cTn concentration in a well characterized population presenting to the ED with symptoms suggestive of acute coronary syndrome for a composite acute cardiac outcome (similar to MACE but with additional cardiac outcomes; see ClinicalTrials.gov Identifier: NCT01994577) (25). In our analyses we found no difference in hs-cTn concentrations between women and men who experienced the composite outcome (25). Extending the analyses now to women 65 years and older (n=378, or 63% of the female population with at least a TIMI score of 1) to see if the LoD cutoff would be useful in this population to prevent misclassification we observed that <4% of women in this group had hs-cTn concentrations at presentation < LoD cutoff (see Figure 1). These data highlight the fact that despite gains in ruling-out patients for MI using the LoD, more sophisticated approaches are needed for patients who are at higher risk and who present with atypical symptoms before efficiency and safety in this setting can be realized.
Acknowledgments
Funding provided by the Canadian Institutes of Health Research with high-sensitivity cardiac troponin tests provided by Roche Diagnostics and Abbott Diagnostics.
Footnote
Provenance and Peer Review: This article was commissioned and reviewed by the Executive Editor Dr. Bing Gu, MD (Department of Laboratory Medicine, the Affiliated Hospital of Xuzhou Medical University, Xuzhou, China).
Conflicts of Interest: Dr. Kavsak has received grants/reagents/consultant/advisor/honoria from Abbott Laboratories, Abbott Point of Care, Abbott Diagnostics Division Canada, Beckman Coulter, Ortho Clinical Diagnostics, Randox Laboratories, Roche Diagnostics and Siemens Healthcare Diagnostics. McMaster University has filed patents with Dr. Kavsak listed as an inventor in the acute cardiovascular biomarker field. Dr. Worster has no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Roffi M, Patrono C, Collet JP, et al. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: Task Force for the Management of Acute Coronary Syndromes in Patients Presenting without Persistent ST-Segment Elevation of the European Society of Cardiology (ESC). Eur Heart J 2016;37:267-315. [Crossref] [PubMed]
- Carlton EW, Khattab A, Greaves K. Identifying Patients Suitable for Discharge After a Single-Presentation High-Sensitivity Troponin Result: A Comparison of Five Established Risk Scores and Two High-Sensitivity Assays. Ann Emerg Med 2015;66:635-645.e1. [Crossref] [PubMed]
- Pickering JW, Than MP, Cullen L, et al. Rapid Rule-out of Acute Myocardial Infarction With a Single High-Sensitivity Cardiac Troponin T Measurement Below the Limit of Detection: A Collaborative Meta-analysis. Ann Intern Med 2017;166:715-24. [PubMed]
- Chapman AR, Lee KK, McAllister DA, et al. Association of High-Sensitivity Cardiac Troponin I Concentration With Cardiac Outcomes in Patients With Suspected Acute Coronary Syndrome. JAMA 2017;318:1913-24. [Crossref] [PubMed]
- Shortt C, Ma J, Clayton N, et al. Rule-in and rule-out of myocardial infarction using cardiac troponin and glycemic biomarkers in patients with symptoms suggestive of acute coronary syndrome. Clin Chem 2017;63:403-14. [Crossref] [PubMed]
- Kavsak PA, Shortt C, Ma J, et al. A laboratory score at presentation to rule-out serious cardiac outcomes or death in patients presenting with symptoms suggestive of acute coronary syndrome. Clin Chim Acta 2017;469:69-74. [Crossref] [PubMed]
- Pickering JW, Greenslade JH, Cullen L, et al. Validation of presentation and 3 h high-sensitivity troponin to rule-in and rule-out acute myocardial infarction. Heart 2016;102:1270-8. [Crossref] [PubMed]
- Pickering JW, Greenslade JH, Cullen L, et al. Assessment of the European Society of Cardiology 0-Hour/1-Hour Algorithm to Rule-Out and Rule-In Acute Myocardial Infarction. Circulation 2016;134:1532-41. [Crossref] [PubMed]
- Worster A, Kavsak P. High-STEACS Algorithm missed fewer patients with acute MI than the ESC Pathway in the ED. Ann Intern Med 2017;167:JC34. [Crossref] [PubMed]
- Worster A, Kavsak P. 4 hs-cTnI algorithms had high sensitivity and low failure rates for ruling out acute MI in the ED. Ann Intern Med 2017;167:JC35. [Crossref] [PubMed]
- Reichlin T, Hochholzer W, Bassetti S, et al. Early diagnosis of myocardial infarction with sensitive cardiac troponin assays. N Engl J Med 2009;361:858-67. [Crossref] [PubMed]
- Kavsak PA, Wang X, Ko DT, et al. Short- and long-term risk stratification using a next-generation, high-sensitivity research cardiac troponin I (hs-cTnI) assay in an emergency department chest pain population. Clin Chem 2009;55:1809-15. [Crossref] [PubMed]
- Kavsak PA, Worster A, Hill SA, et al. Analytical comparison of three different versions of a high-sensitivity cardiac troponin I assay over 10 years. Clin Chim Acta. 2017;475:51-5. [Crossref] [PubMed]
- Herman DS, Kavsak PA, Greene DN. Variability and Error in Cardiac Troponin Testing: An ACLPS Critical Review. Am J Clin Pathol 2017;148:281-95. [Crossref] [PubMed]
- Than M, Herbert M, Flaws D, et al. What is an acceptable risk of major adverse cardiac event in chest pain patients soon after discharge from the Emergency Department?: a clinical survey. Int J Cardiol 2013;166:752-4. [Crossref] [PubMed]
- Kavsak PA, Worster A. Dichotomizing high-sensitivity cardiac troponin T results and important analytical considerations. J Am Coll Cardiol 2012;59:1570. [Crossref] [PubMed]
- Kavsak PA, Hill SA, McQueen MJ, et al. Implications of adjustment of high-sensitivity cardiac troponin T assay. Clin Chem 2013;59:574-6. [Crossref] [PubMed]
- Kavsak PA, Clark L, Jaffe AS. Effect of Repeat Measurements of High-Sensitivity Cardiac Troponin on the Same Sample Using the European Society of Cardiology 0-Hour/1-Hour or 2-Hour Algorithms for Early Rule-Out and Rule-In for Myocardial Infarction. Clin Chem 2017;63:1163-5. [Crossref] [PubMed]
- Lyon AW, Kavsak PA, Lyon OA, et al. Simulation Models of Misclassification Error for Single Thresholds of High-Sensitivity Cardiac Troponin I Due to Assay Bias and Imprecision. Clin Chem 2017;63:585-92. [Crossref] [PubMed]
- Kavsak PA, Worster A, Oliver R, et al. Variability between reagent lots for high-sensitivity cardiac troponin I may affect performance of early-rule out strategies. Can J Cardiol 2017; [Crossref]
- Cullen L, Mueller C, Parsonage WA, et al. Validation of high-sensitivity troponin I in a 2-hour diagnostic strategy to assess 30-day outcomes in emergency department patients with possible acute coronary syndrome. J Am Coll Cardiol 2013;62:1242-9. [Crossref] [PubMed]
- Carlton EW, Pickering JW, Greenslade J, et al. Assessment of the 2016 National Institute for Health and Care Excellence high-sensitivity troponin rule-out strategy. Heart 2017; [Epub ahead of print]. [Crossref] [PubMed]
- Shah AS, Griffiths M, Lee KK, et al. High sensitivity cardiac troponin and the under-diagnosis of myocardial infarction in women: prospective cohort study. BMJ 2015;350:g7873. [Crossref] [PubMed]
- Rubini Giménez M, Twerenbold R, Boeddinghaus J, et al. Clinical Effect of Sex-Specific Cutoff Values of High-Sensitivity Cardiac Troponin T in Suspected Myocardial Infarction. JAMA Cardiol 2016;1:912-20. [Crossref] [PubMed]
- Kavsak PA, Worster A, Shortt C, et al. High-sensitivity cardiac troponin concentrations at emergency department presentation in females and males with an acute cardiac outcome. Ann Clin Biochem 2017; [Epub ahead of print]. [PubMed]
Cite this article as: Kavsak PA, Worster A. Does the LoD cutoff + the TIMI score = a NICE approach to rule-out a major adverse cardiac event in the emergency department? J Lab Precis Med 2017;2:92.


